🔍 Background: Why Dry Electrodes are the “Holy Grail” of Manufacturing
Traditional wet-slurry coating is limited by the drying rates of solvents (e.g., NMP) and binder migration, creating a physical “ceiling” for electrode mass loading. In contrast, Dry Electrode Technology (DET) completely bypasses solvent usage, utilizing fibrillated binders (such as PTFE) to construct self-supporting membranes.
Core Advantages:
- ➡️ Ultra-High Areal Capacity: Easily achieves 5–12 mAh/cm², a threshold nearly impossible for wet coating.
- ➡️ Superior Structural Stability: Eliminates micro-pores and cracks caused by solvent evaporation, enhancing the contact intimacy of active materials.
- ➡️ Process Simplification: Removes expensive coaters and solvent recovery systems, significantly reducing energy consumption.
📊 Empirical Deconstruction: Hardcore Electrochemical Data of Multi-System Dry Electrodes
By preparing dry electrodes for various cathode systems and matching them with coin cell testing, we obtained compelling first-cycle data:
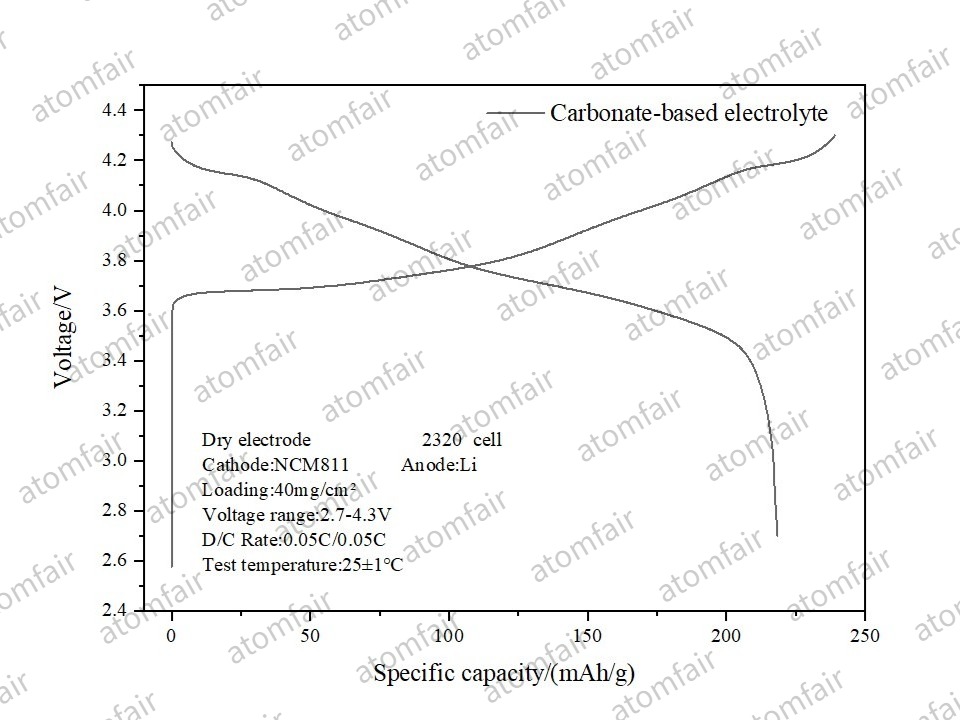
1. High-Nickel NCM811: Challenging High-Loading Limits
At a high loading of 40 mg/cm², the dry NCM811 electrode released a specific capacity of 218.39 mAh/g with an Initial Coulombic Efficiency (ICE) of 91.37%. This proves that dry processing maintains excellent electrical network connectivity even at high specific energies.
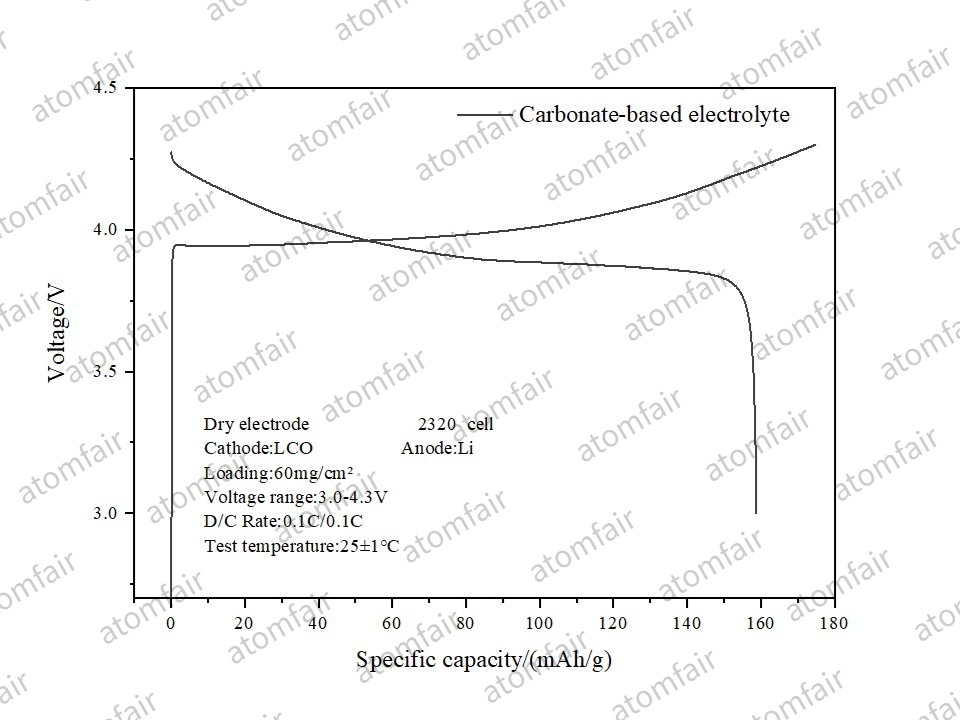
2. Lithium Cobalt Oxide (LCO): The Pinnacle of Thick Electrodes
Under an extreme loading of 60 mg/cm², the dry LCO electrode maintained a specific capacity of 158.8 mAh/g and an ICE of 90.85%. For 3C batteries pursuing ultimate volumetric energy density, this ultra-thick electrode technology is a landmark achievement.
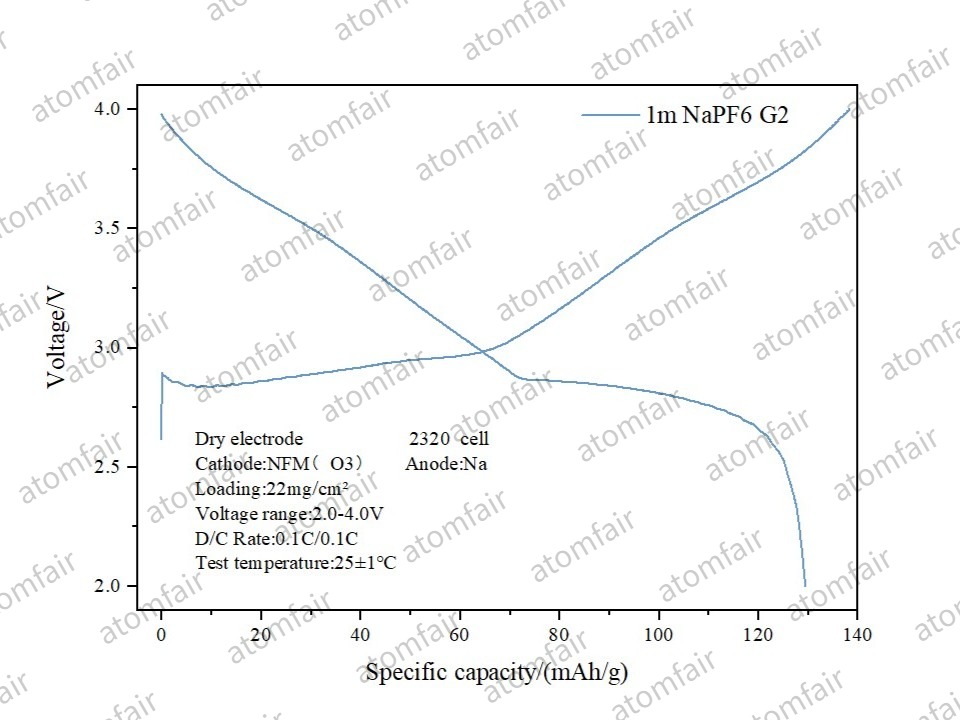
3. Sodium-ion (NFM) System: Universal Applicability
In the sodium-ion NFM system, dry processing achieved a loading of 22 mg/cm² with a specific capacity of approx. 130 mAh/g, demonstrating high process maturity beyond lithium-ion applications.
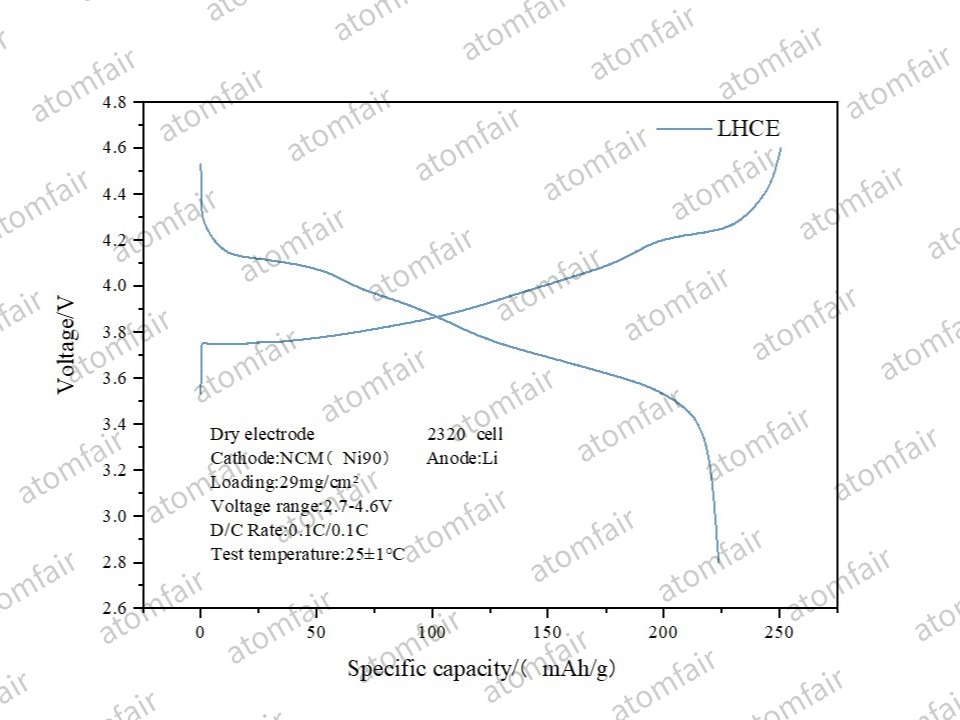
4. Ni90 System: Challenging 4.6V Ultra-High Voltage
This was the standout result of the evaluation. Within a wide voltage range of 2.7–4.6V, the dry Ni90 electrode achieved a specific capacity of approximately 240 mAh/g.
- Insights: Ni90 is prone to gas evolution at high voltages. Because dry processing leaves no solvent residue, the interface is “cleaner,” effectively reducing side reactions during the initial cycle.
🛠️ Technical Specifications Benchmarking (Dry Process Measured Values)
| Material System | Areal Loading (mg/cm²) | Specific Capacity (mAh/g) | Initial Efficiency (ICE) |
| High-Nickel NCM811 | 40 | 218.39 | 91.37% |
| Lithium Cobalt Oxide (LCO) | 60 | 158.80 | 90.85% |
| High-Nickel Ni90 | 29 | ~230 | —— |
| Sodium-ion (NFM) | 22 | ~130 | —— |
💡 Deep Insight: How Dry Processing Reshapes Performance
- Fibrillated Network Support: The PTFE-formed network structure, generated under shear force, imparts high mechanical strength to the electrode without sacrificing ion transport channels—ideal for ultra-thick electrodes.
- Areal Capacity Breakthrough: Since solvent diffusion is not an issue, electrode thickness is virtually unlimited, providing the fundamental logic for achieving 5–12 mAh/cm² areal capacities.
- Customized Optimization: For high-activity surfaces like high-nickel cathodes, the absence of solvent avoids parasitic reactions, moving the interface toward a state of thermodynamic stability.
🔬 Advanced R&D Roadmap for Laboratories
If you are exploring next-generation high-loading electrodes or solid-state battery processes:
- System Benchmarking: We recommend using our Customized Dry Electrode Samples (LCO, NCM, NFM) as your experimental baseline to quickly validate electrolyte or anode compatibility.
- Dry Process Auxiliaries: We provide specialized PTFE binders and matching conductive agents optimized for dry processing, helping your research group bridge the “loading gap” of traditional wet coating.