🌐 Foreword: The Final Push from Lab to 500 Wh/kg
Lithium Metal Batteries (LMBs) are regarded as the core technology for breaking the 500 Wh/kg energy density threshold due to the extreme theoretical specific capacity of lithium (3,860 mAh/g) and its ultra-low electrode potential. However, unlike the low-loading models common in laboratories, achieving 4.6V ultra-high voltage operation under industrial-grade mass loading (24 mg/cm²) makes electrolyte stability the ultimate determinant of system survival.
This issue shares the empirical performance of the NCM811 || Li system at the limits of energy density, demonstrating how perfluorinated electrolytes “tame” high-nickel cathodes under high-voltage stress.
📊 Performance Deconstruction: Authenticity at 24 mg/cm² Industrial Loading
This evaluation utilized 1.5 Ah pouch full cells to conduct an in-depth assessment within a 2.5V – 4.6V limit-testing window:
1. Massive Specific Capacity Utilization
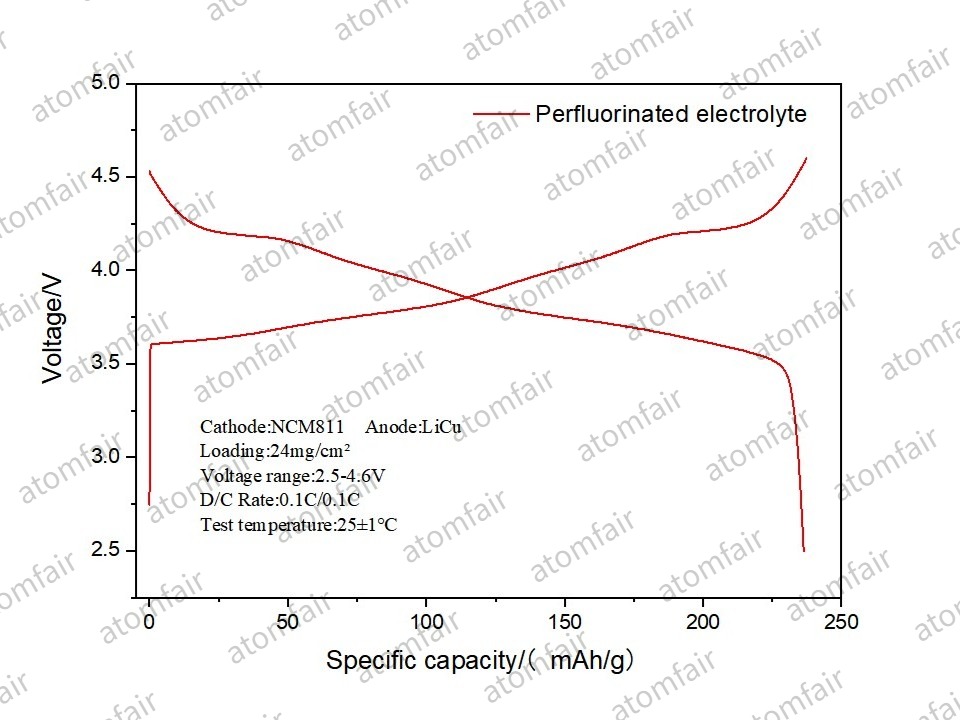
- Core Data: Within an NCM811 system featuring a single-sided loading of 24 mg/cm², the material achieved a specific capacity of 236.37 mAh/g.
- Energy Dimension: The measured cell energy density reached 463.13 Wh/kg.
- Insights: The Galvanostatic Charge-Discharge (GCD) curves show that even at a 4.6V ultra-high voltage, the plateaus remain distinct and stable. This indicates that high-nickel NCM811 retains excellent kinetic activity even in a state of deep delithiation.
2. Interfacial Regulation of Cycle Life
The most severe challenges for high-voltage NCM811 are electrolyte oxidation at the cathode surface and dendrite growth on the lithium metal anode.
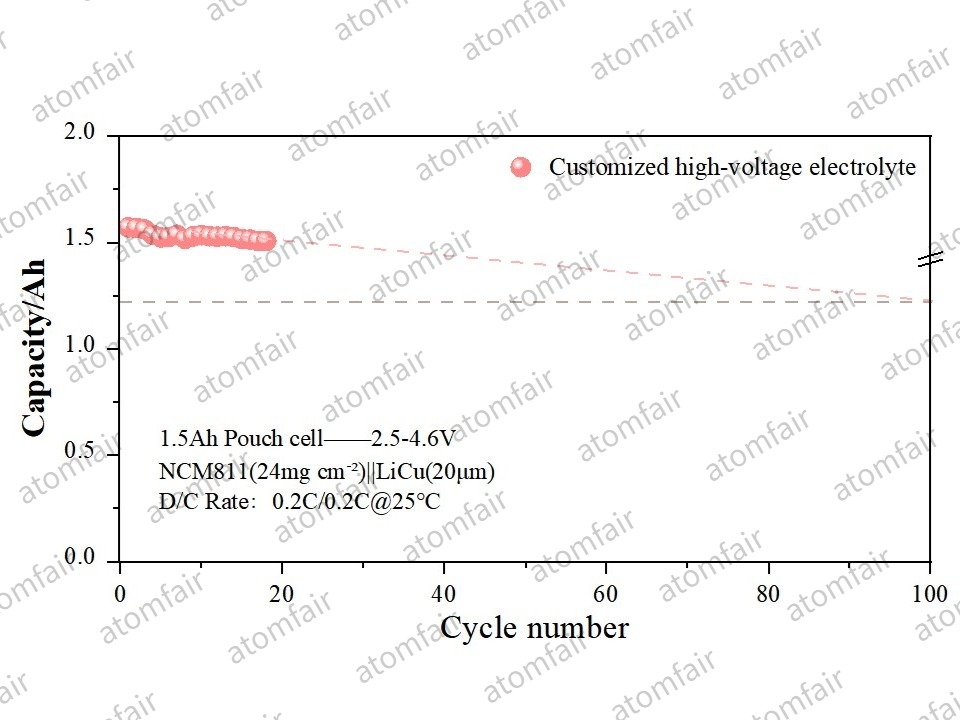
- Measured Data: Paired with our Customized Fluorinated/Perfluorinated Electrolyte, the system maintained a capacity retention of 96.50% after 20 cycles.
- Trend Projection: Based on the current capacity decay slope, this industrial-grade system is projected to achieve a cycle life of 100 cycles while maintaining 80% capacity retention.
💡 Deep Insight: How Perfluorinated Electrolytes Safeguard the 4.6V Interface
In a strongly oxidative 4.6V environment, conventional electrolytes fail rapidly on the NCM811 surface, leading to gas evolution and surging resistance.
- Construction of Anti-Oxidative CEI: By lowering the HOMO energy levels of the solvent molecules, the customized fluorinated electrolyte induces the formation of a robust, LiF-rich Cathode Electrolyte Interphase (CEI) on the cathode side. This layer effectively isolates high-valence nickel ions from attacking the solvent, significantly suppressing interfacial side reactions.
- Synergistic Anode Protection: The perfluorinated components protect not only the cathode but also participate in forming a dense Solid Electrolyte Interphase (SEI) on the lithium metal anode. This slows dendrite growth and active lithium loss, which is critical for maintaining cycling stability in large 1.5 Ah-class cells.
🛠️ Technical Specifications Benchmarking
- Cathode System: Industrial High-Loading NCM811 (24 mg/cm²)
- Anode System: Composite Lithium-Copper (LiCu, 20µm)
- Cut-off Voltage: 2.5V – 4.6V
- Core Solution: Perfluorinated Electrolyte (Customized 4.6V Grade)
- Measured Energy Density: 463.13 Wh/kg (1.5 Ah Pouch Cell)
🔬 Laboratory Selection Guidance
If you are currently exploring the engineering of high-energy-density lithium metal batteries:
- Bypass the “Low-Loading” Trap: Failure modes in pouch cells are often difficult to replicate with laboratory coin cell data. We recommend using our 24 mg/cm² high-loading electrodes for system-level validation.
- Interfacial Engineering Benchmark: Electrolyte stability at 4.6V is at the forefront of battery research. Utilizing our Perfluorinated Electrolyte Series as a benchmark can help research groups quickly identify interfacial mechanisms and accelerate high-level academic publications.