🔍 Background: The High-Voltage Temptation and Dilemma of LCO
Since its inception by John B. Goodenough, Lithium Cobalt Oxide (LCO) has dominated the consumer electronics market (smartphones, laptops, etc.) due to its ultra-high tap density (>4 g/cm³) and stable discharge plateau. To meet the demand for longer battery life, pushing the cut-off voltage from 4.2V to 4.5V and beyond is an inevitable industry trend.
However, at high voltages, LCO suffers from severe bulk structural collapse (phase transitions) and violent electrolyte oxidation at the cathode surface. How can we preserve cycle life while increasing capacity? This issue shares an industrial-grade optimization solution for the LCO-Graphite (Gr) system.
📊 Empirical Deconstruction: High-Energy Performance Under Industrial Loading
This evaluation utilized industrial-grade pouch full cells to challenge performance limits within a 2.5V – 4.5V window:
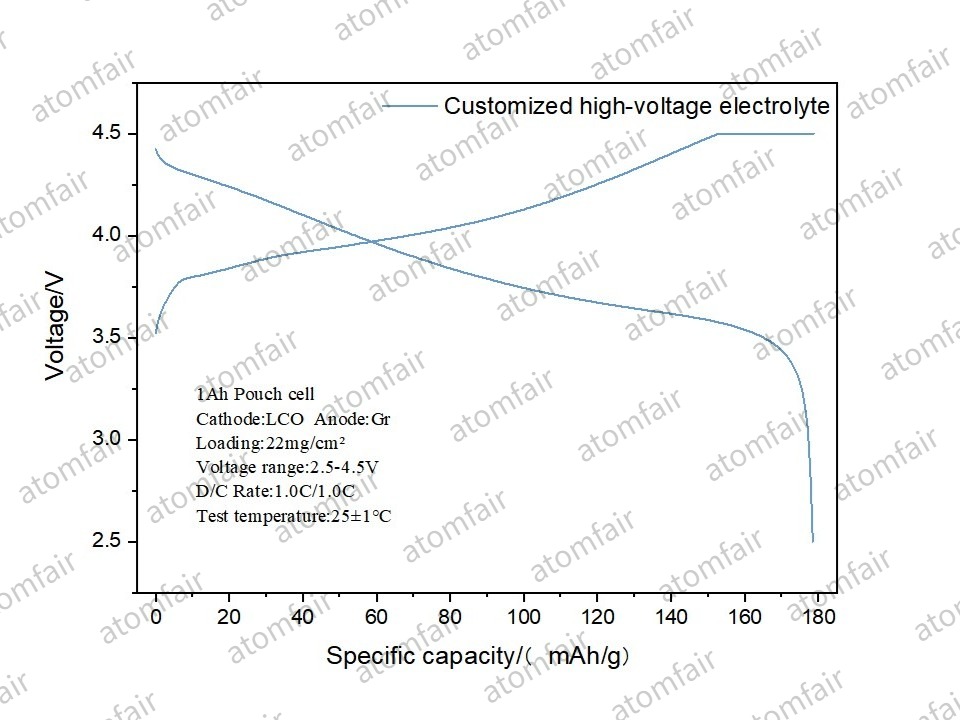
- ➡️ Specific Capacity Utilization: The measured specific capacity reached 178.45 mAh/g, representing a significant energy density leap compared to conventional voltage systems.
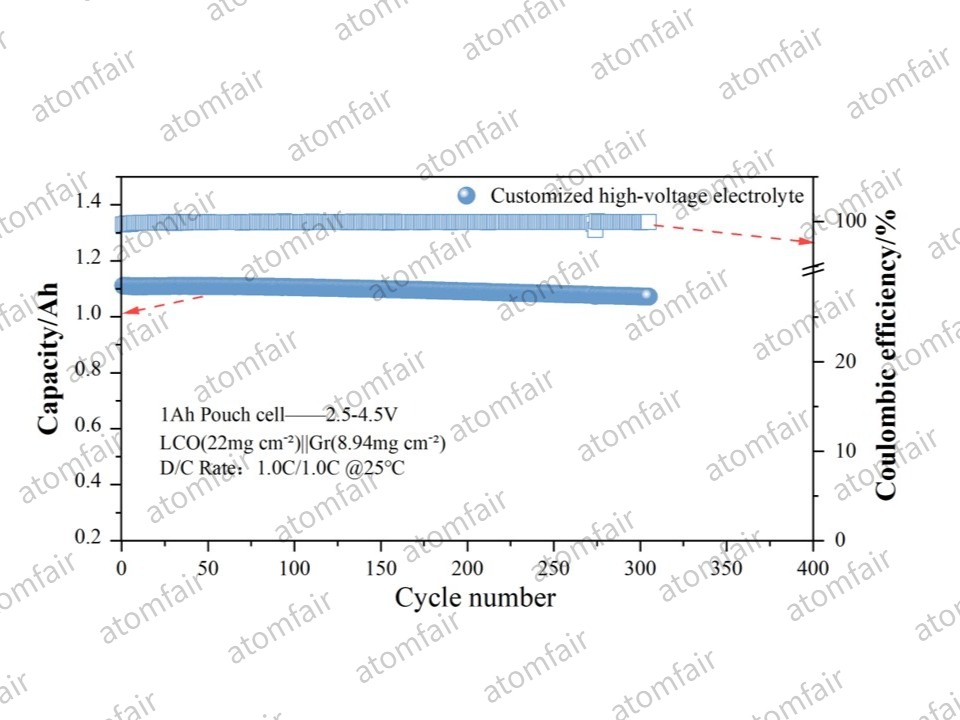
- ➡️ Extreme Cycling Stability: As shown in the cycling data, paired with a Customized High-Voltage Electrolyte, the cell maintained a capacity retention of 96.47% after 307 cycles.
- ➡️ Coulombic Efficiency (CE): The consistently high CE stability throughout the process (see blue scatter plots) indicates that this cell is on track to achieve an ultra-long lifespan exceeding 1,500 cycles under industrial standards.
💡 Deep Insight: Dual Synergy of Materials and Interfaces
The key to achieving a stable 4.5V cycle lies in resolving both “internal and external” stressors at high voltages:
- Bulk Phase Stability: By utilizing High-Voltage Modified LCO, elemental doping and surface coatings effectively suppress the dissolution of cobalt (Co) ions and the evolution of lattice oxygen at high potentials.
- Dynamic Interfacial Repair: The Customized High-Voltage Electrolyte induces the formation of a dense Cathode Electrolyte Interphase (CEI) on the LCO surface. This layer effectively blocks continuous attacks on solvent molecules by highly active cobalt ions, ensuring interfacial kinetics even under a high mass loading of 22 mg/cm².
🛠️ Technical Specifications Benchmarking
| Key Indicators | Empirical Data |
| Cathode System | High-Voltage LCO (Single-sided 22 mg/cm²) |
| Anode System | High-Performance Artificial Graphite (Single-sided 8.94 mg/cm²) |
| Voltage Window | 2.5V – 4.5V |
| Specific Capacity | 178.45 mAh/g |
| Cycling Performance | 96.47% @ 307 Cycles (Projected >1,500 Cycles) |
🔬 Advanced R&D Roadmap for Laboratories
If your primary research focuses on high-energy-density consumer electronics batteries:
- Engineering Benchmarking: We recommend using the LCO-Gr Comprehensive Material Solution presented in this evaluation as a baseline to eliminate experimental errors caused by material incompatibility under high-voltage stress.
- Interfacial Mechanism Exploration: This system serves as an ideal model for investigating “high-voltage cathode failure” and the “development of multi-functional additives.”